Efficacy and Safety of Sofosbuvir with Simeprevir in Hepatitis C Infected Patients with Severe Chronic Kidney Disease: A Systematic Review and Meta-Analysis
DOI:
https://doi.org/10.5530/BEMS.2016.1.2Keywords:
Chronic kidney insufficiency, Efficacy, Hepatitis C, Sofosbuvir, Simeprevir, SafetyAbstract
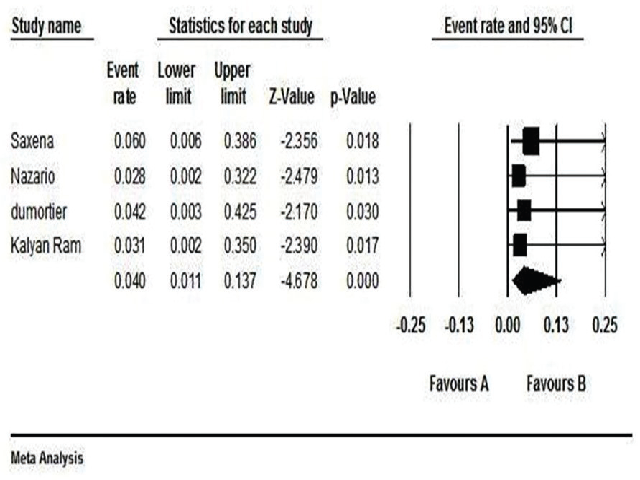
Background & Aims: Conventional treatment (interferon or ribavirin) for Hepatitis C viral (HCV) infection in patients with severe chronic kidney disease (CKD) has limitations of high dropout and less response rate. Directly acting antivirals raise hopes for HCV treatment in these patients. This meta-analysis was performed to evaluate the evidence for efficacy and safety of sofosbuvir and simeprevir, with or without ribavirin, in HCV-infected patients with severe CKD. Methods: Data was collected from Medline database, clinical-trial registry sites, and conference proceedings. This meta-analysis screened 78 studies. Quality of studies was assessed by New-Castle Ottawa scale. Heterogeneity and publication bias was checked by chi-square Q test and Eggers’ test, respectively. Summary estimate of SVR12 and dropout rate was calculated at 95% confidence interval (CI). Results: Seven relevant clinical studies were identified. Two case-series and one case-report were excluded; only four studies were eligible for analysis. Three studies were cohort and one was retrospective-cohort in nature. Data was analyzed for 56 subjects. 33/56 subjects had cirrhosis. 39/56 subjects were on hemodialysis. 37/56 subjects were male. 30/56 subjects were treatment-naive. Pooled estimate of SVR12 was found 0.897 (CI 95%=0.957-0.772; p<0.01) and dropout estimate was 0.040 (CI 95%=0.011-0.137; p<0.01). Only one subject discontinued the treatment due to worsening Renal function regardless of ribavirin. Conclusion: This study concluded that combination of sofosbuvir and simeprevir, with or without ribavirin, was significantly effective and safe in HCV patients with severe CKD. For conclusive results, more data is required as this study involved only limited number of subjects.